FACILITIES
High-tech facilities,
hot concerns
The medical research facilities cover more than 2,000 m² spread over our 2 sites in Alken and Diepenbeek. In total we have 11 fully equipped examination rooms, an intensive monitoring and treatment department with 12 individual bedrooms, 2 on-site GMP compliant storage rooms for medication and 2 specialized labs for processing and storing biological samples, 2 fully equipped Polysomnography (PSG) facilities and meeting rooms for monitoring, initiation and close-out visits.
In addition, since 2022, Anima Research Center has also had a structural collaboration with Jessa Hospital to conduct early phase and patient studies together (day & night stays) and this includes support from Jessa's lab, pharmacy, and intensive care services with our medical team as admitted physicians.

Day and night accommodation
12 treatment rooms with high-tech video, sound, light, humidity and temperature monitoring.

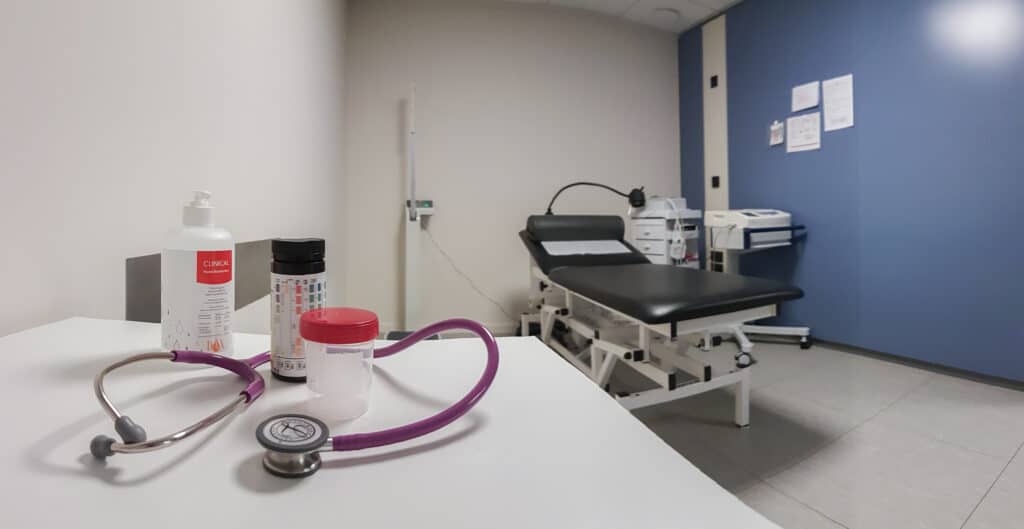
Research spaces
Performing physical examination,
measurement of vital parameters, 12-lead ECG, etc.

Access control
Full batch controlled access to all rooms with specific delegation according to function and permanent registration.

Meeting possibilities
Offices (6) and meeting rooms (3) for initiation, monitoring and close-out visits as well as audits with the pharmaceutical companies, strictly separated from research work with participants to ensure anonymity.

Waiting and relaxation areas
Spacious sitting room with kitchenette and sofa 2 archives with strict electronic access control. Continuous temperature and humidity monitoring as well as strict electronic access control.

Lab GLP compliant
Processing and storage of biological samples with continuous temperature and humidity monitoring according to the international Good Lab Practice standard.

Pharmacy: GDP-compliant
Rooms fully equipped in accordance with international Good Distribution Practice (GDP). This is where the experimental medication is stored in accordance with the very specific safety procedures of the Federal Agency for Medicines and Health Products (FAMHP) and allocated to the study participant. These strictly secured medication rooms are certified by the FAMHP.

Medical Permanence
Since the Anima Research Center is located on the private campus of the family Dr. Erik Buntinx - Dr. Hilde Bollen, there is always the direct presence of at least one physician within walking distance when a study participant is present.
Accessibility is further assured by an internal electronic telephone system and a specific professional WIFI network that is active across the entire campus and in all its buildings. Thus, every study participant is assured of the necessary immediate medical intervention.
This is where the Anima Research Center distinguishes itself significantly.
An important guarantee for the participant, the sponsor and the supervising authorities.